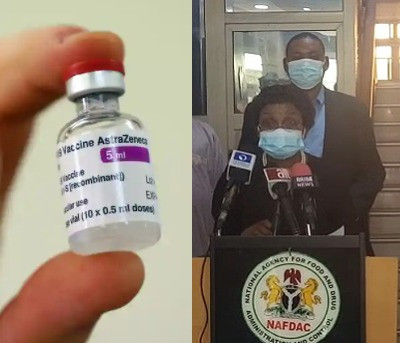
The National Agency for Food and Drug Administration and Control in Nigeria, NAFDAC, has approved the Oxford/AstraZeneca COVID19 vaccine for use in Nigeria.
At a press conference held this morning, February 18, the Director-General of NAFDAC, Professor Mojisola Adeyeye, says the agency got the vaccine’s dossier a week ago the NAFDAC safety committee went to work immediately to evaluate its safety and efficacy for Nigerians.
Adeyeye said that the vaccine could be stored at 2 to 8-degree centigrade. According to the DG, there are three additional vaccines undergoing evaluation. Still, the evaluation on Astrazeneca shows that the vaccine is effective against the virus’s UK variant, which has been reported in Nigeria. She disclosed that the South African variant has not been reported in Nigeria and that the agency has over 30 herbal medicine undergoing review for listing.
The Oxford/AstraZeneca vaccine, also known as ChAdOx1 to-19, or AZD1222, is a viral vector vaccine. Scientists used an adenovirus, originally derived from chimpanzees, and modified it to train the immune system to mount a strong response against SARS-CoV-2 (the virus that causes COVID-19).
Nigeria is expected to get the first batch of the COVID19 vaccines by the end of February.











































Leave a comment